Immune monitoring and Medical Immunology (LUMC Strategic Fund 2019-2-040 & EuroFlow LSHB-CT-2006-018708)
Lead: Mihaela Zlei and Jacques J.M. van Dongen
To support translational research projects, diagnostics and clinical studies, continuous effort is made to develop, optimize and standardize new immune monitoring techniques. For example, by mapping the immune system more accurately with molecular and cellular techniques, the disease status and the effects of a treatment can be investigated with increasing precision. Innovation within these and other research lines within the immune monitoring program therefore directly contributes to translational research.
Important areas of application include diseases of the immune system and immunotherapies in a large number of medical fields. Therefore, many collaborations with clinical departments of LUMC are included in this research line. In addition, immune monitoring is used for clinical studies in collaboration with pharmaceutical industry.

In order to ensure high-quality and reproducible research, and to support the implementation of immune monitoring techniques in laboratories around the world, standardization of these techniques in an international, multi-center context is a priority,such as within the EuroFlow and EuroClonality research consortia (established under Grant LSHB-CT-2006-018708 and BMH4-CT98-3936 of the Sixth and Fourth Framework Programme (FP6 and FP4) of the European Commission, respectively). The LUMC immune monitoring team is continuously contributing to these research consortia (see the EuroFlow and EuroClonality websites).
Prof. Jacques J.M. Van Dongen is the founding chairman of EuroClonality (1996-2011) and the founding chairman of EuroFlow (since 2006).
Monitoring of cancer via tissue macrophages (TiMaScan: ERC Advanced Grant ERC-2015-AdG-695655)
Lead: Cristina Teodosio, Paula Díez, Indu Khatri and Jacques J.M. van Dongen
PhD students: Wouter B.L. van den Bossche, Kyra van der Pan, Anniek de Jager

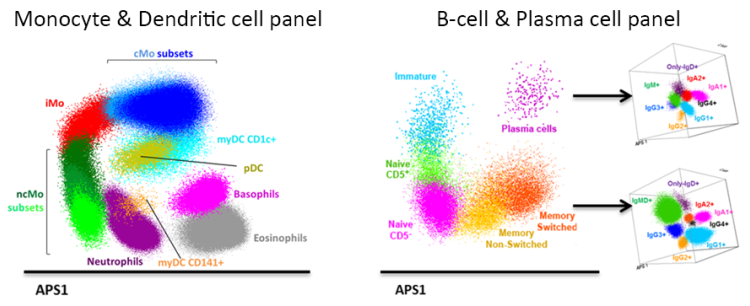
The increasing knowledge, obtained in the past decades, on the biology and function of monocytes, macrophages and dendritic cells, revealed key functions for these cells in maintenance of homeostatic balance that go beyond antigen presentation. Their ability to monitor and sense micro-environmental changes, and perform tissue clean-up tasks, make these cells ideal candidates for assessment of disturbances in homeostasis. In line with this, several studies have shown the relevance of monitoring myeloid antigen presenting cells in patient care settings. Nevertheless, despite the extensive research on the monocyte-macrophage system, the usefulness of evaluating these populations is still limited due to lack of knowledge of the precise nature of the monocyte-macrophage maturation pathway and their relation with dendritic cell populations across the different tissues.
The TiMaScan research line aims at a truly translational research, by focusing on the understanding the processes involved in the differentiation of the monocytic lineage and their relation with dendritic cells, as a basis for development of new, standardized tools to be implemented in patient care. In order to do so, we employ multi-omics approaches, including transcriptomics, proteomics and high-end flow and mass cytometry, for phenotypic and functional evaluation of the monocyte-macrophage system across different body compartments, in both steady-state and in response to distinct “stressors” such as cancer, infection, vaccination or trauma.
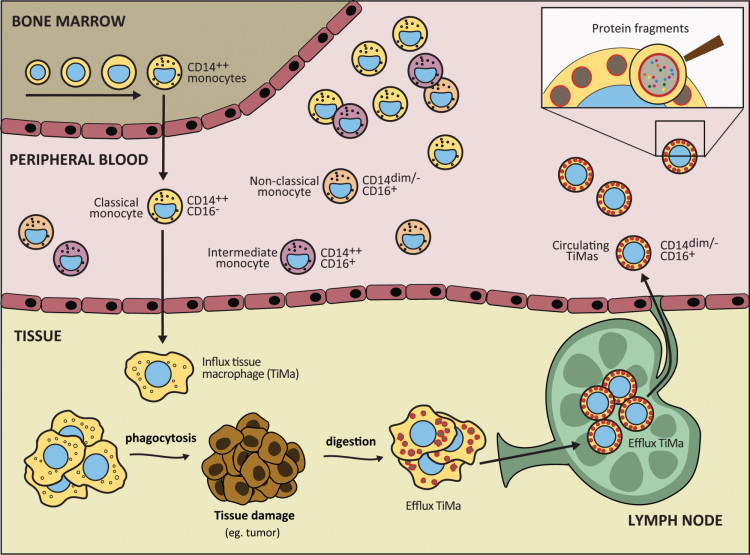
More specifically, we apply the knowledge obtained from the dissection of the monocyte-macrophage system maturation pathway (particularly regarding the recirculation of macrophages from tissues via the lymph system to the blood stream), phagocytosis and digestion processes, for the design of an innovative, minimally-invasive diagnostic approach for early detection of cancer cells. This is based on the novel concept (Figure 2) that tissue macrophages (TiMas) continuously phagocytize and digest apoptotic cells (particularly in case of cancer) and subsequently recirculate to the blood stream, where the TiMas can be detected and scanned for their phagolysosomal contents (TiMaScan), using the novel EuroFlow-based advanced flow cytometric techniques.
Our research is performed in close collaboration with local partners (e.g. Depts. of Internal Medicine, Surgery, Pathology, Cardiology, Pediatrics, etc.), national partners (e.g. Erasmus Medical Center, Martini Hospital Groningen), and international partners (e.g. University of Salamanca, Spain).

More information about the TiMaScan project (ERC Advanced Grant ERC-2015-AdG-695655) can be found on the TiMaScan website.
Monitoring of the immune response after vaccination
Lead: Magda Berkowska, Indu Khatri and Jacques J.M. van Dongen
PhD students: Annieck Diks, Lisanne Overduin
In this research line, immune monitoring is used to gain better insights into the immune response after vaccination and to use this knowledge to understand and improve the effectiveness and safety of vaccinations, i.e. to understand the mechanisms of protection, formation and waning of the immune response to vaccines.
Vaccination, or induction of an immune response against a pathogen by the administration of the weakened or dead pathogen or of isolated antigens, causes a complex dynamic change in the immune system. Many different cell populations are involved in this process. By visualizing the different populations of immune cells and their changes over time, we can learn more about the involvement of these populations in an immune response. With this knowledge we can better predict the effectiveness and safety of vaccinations and contribute to the development of more effective vaccination strategies in the future.
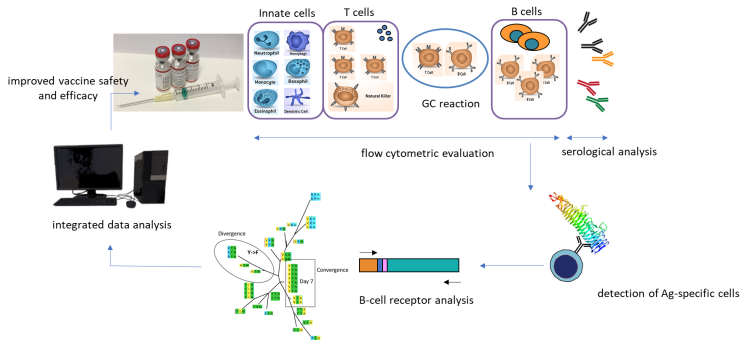
A large project within this research line is PERISCOPE (PERtussIS Correlates Of Protection Europe; Innovative Medicines Initiative (IMI) Grant 115910). This project focuses on vaccination against Bordetella pertussis (whooping cough), and aims to accelerate the development of a more effective and safer vaccine against this disease by identifying long-term biomarkers of immunity. Immune monitoring techniques are developed and applied to map the response of the immune system to vaccination against or infection with Bordetella pertussis. One example is the development of antibody panels for flow cytometry with the aim to identify large numbers of subpopulations of e.g. monocytes, T cells and B cells. In addition, antigen-specific B cells are examined in more details on the basis of single cell RNA sequencing to understand the selection processes in antigen-specific antibody repertoire. We develop bioinformatic tools to better describe the processes of convergent and divergent evolution of vaccine-specific cells.

The PERISCOPE Consortium is supported by the European Innovative Medicines Initiative (IMI) and the Bill & Melinda Gates Foundation. More information about the PERISCOPE project can be found on the PERISCOPE website.
In addition to studies on immune responses to Bordetella pertussis, we broaden our research scope by several collaborations within and outside of the LUMC. Currently, we support a study on immune responses to Human Papillomavirus (conducted at the National Institute for Public Health and the Environment, RIVM) and develop new strategies to monitor immune responses to rabies vaccine (together with the Department of infectious Diseases, LUMC). We hope to implement our tools to improve vaccination safety and efficacy in immunocompromised individuals.


